-

K-Clip® Transcatheter Tricuspid Annuloplasty System
K-Clip® is the world's first transcatheter tricuspid valve annuloplasty system independently developed by Huihe Medical, officially approved for market by the National Medical Products Administration (NMPA) in March 2025. This product is used to treat tricuspid regurgitation, transforming traditional open-chest surgery into a minimally invasive vascular puncture approach with small trauma, fast recovery, excellent efficacy, and simple operation, averaging only 30 minutes per procedure.¥ 0.00Product Details +
-
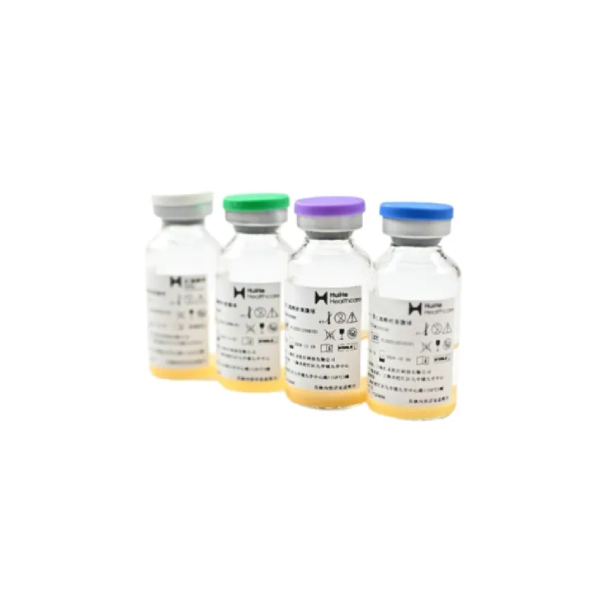
Vispearl® PVA Embolic Microspheres
Vispearl® is a radiopaque embolic microsphere with independent intellectual property rights. It can be visualized under X-ray, carries tumor drugs, and is suitable for TACE in the treatment of liver cancer.¥ 0.00Product Details +
-
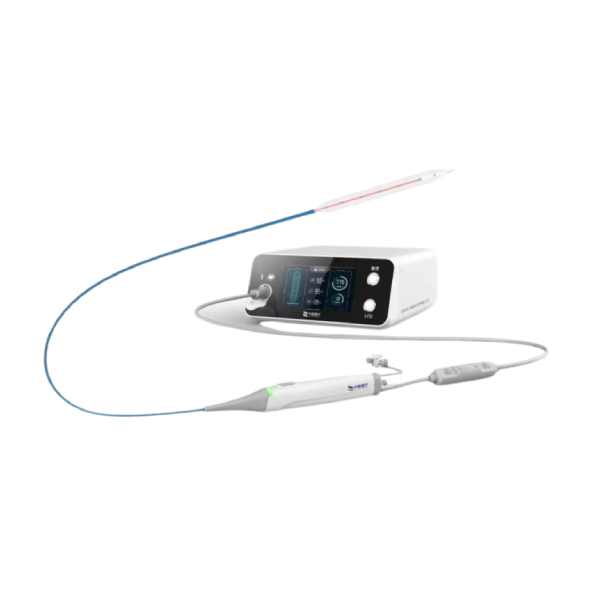
C-Wave® Peripheral Intravascular Lithotripsy (IVL) Catheter System (CP)
The C-Wave® Peripheral Intravascular Lithotripsy (IVL) Catheter System, independently developed by Zhonghui Medical—a subsidiary of H&H Medical—is indicated for the treatment of intravascular calcified lesions in peripheral arteries. It can be used either as a standalone therapy or in combination with stents and other interventional devices.¥ 0.00Product Details +
-

The C-Wave® Coronary Intravascular Lithotripsy Catheter System(CC)
The C-Wave® Coronary Intravascular Lithotripsy Catheter System, independently developed by H&H Medical's subsidiary Zhonghui Medical, is suitable for coronary calcification and occlusive lesions where stents are difficult to pass. This product, led by Professor Chen Shaoliang and Professor Zhang Junjie from Nanjing First Hospital, has obtained the NMPA registration certificate in 2024.¥ 0.00Product Details +
-

S-wan™ Dual-Point Adjustable Curved Sheath
S-wan™ dual-point adjustable bending sheath is the world's first multi-point bending-controlled delivery sheath tube independently developed by H&H Medical. It is suitable for vascular access such as structural cardiac intervention, peripheral intervention and nerve intervention, with a diameter of 6Fr to 23Fr.
This product obtained the National Medical Products Administration (NMPA) registration certificate in 2023.¥ 0.00Product Details +
Consulting Services:question@hh-healthcare.com








