GO ALL IN, DELIVER OUR MISSION
H&H Medical's products have successfully entered the North American and European markets. Going forward, the company will further expand its overseas operations, driving global adoption of China-developed medical devices. Guided by the principles of 'quality first, reliability guaranteed, and customer-centric service,' the company is committed to collaborating with domestic and international partners to shape a brighter future for global healthcare.
SHANGHAI H&H MEDICAL TECHNOLOGY CO., LTD
H&H Medical is a high-tech enterprise specializing in the R&D, manufacturing, and commercialization of cardiovascular interventional medical devices, with its headquarters in Songjiang District, Shanghai. Driven by "Integration of Medical and Engineering Expertise" and "Independent R&D innovation", H&H Medical is dedicated to promoting the globalization of China's intelligent manufacturing of high-end medical devices.
The company holds industry-leading advantages in delivery systems, precision manufacturing, simulation modeling, and automated production. In the fields of transcatheter heart valve interventions, vascular interventions, drug delivery interventions, and smart interventional technologies, H&H Medical has commercialized multiple self-developed, world-leading products.
Founded by Ms. Lin Lin, a former cardiologist at Ruijin Hospital, Shanghai Jiao Tong University School of Medicine, has 20 years of clinical experience and R&D experience.
About Us
PROMOTE THE GLOBALIZATION OF CHINESE ORIGINAL MEDICAL DEVICES.
Product Center
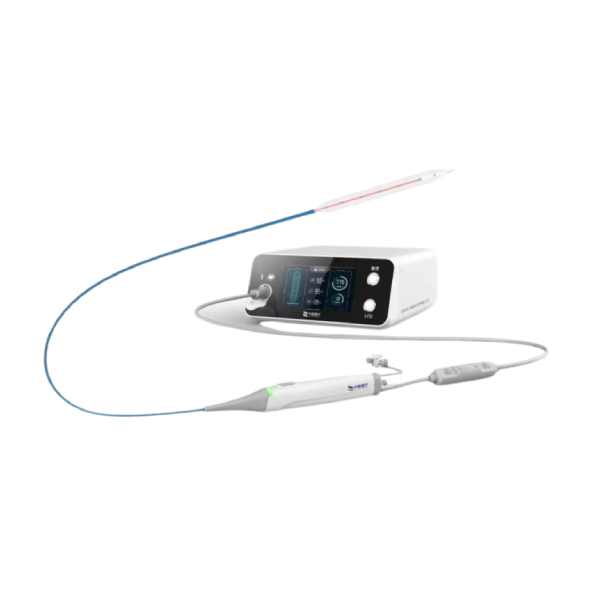
K-Clip ®
Transcatheter Tricuspid Annuloplasty System
The world's first transcatheter tricuspid annuloplasty systemreplicates classic surgical procedures and repairs tricuspid regurgitation through minimally invasive surgery. It was approved for market by the National Medical Products Administration (NMPA) in March 2025. Key advantages include minimally invasive,repaid recovery, effective outcomes,user-friendly design and with an average operation time of just 30 minutes.
K-Clip® fills a critical gap in China's interventional tricuspid valve field, becoming the first certified transcatheter solution for tricuspid regurgitation domestically. This pioneering device also ushers in a new era of Transcatheter Tricuspid Valve Annuloplasty (TTVA) globally, pioneering international research trends in tricuspid valve interventions. With over 250 patients successfully treated in China, K-Clip® has been granted access to the NMPA's priority review pathway for innovative medical devices.
K-Clip® has advanced global clinical validation through successful procedures in the U.S., Europe, and Asia-Pacific, earning recognition in leading cardiovascular journals.
▸ Obtained approval from the National Medical Products Administration (NMPA)
PRODUCT CENTER
Vispearl ®
radiopaque embolic microspheres
PRODUCT CENTER
Developed with independent intellectual property rights, Vispearl® is a breakthrough radiopaque embolization microsphere platform capable of drug loading for tumor therapy. It is indicated for Transarterial Chemoembolization (TACE) in hepatocellular carcinoma (HCC) treatment.
Key Features
• Smallest particle size of 40μm: Enables ultra-precise embolization and a stable release profile.
• Radiopaque visibility under X-ray: Facilitates real-time imaging guidance, traceability, and synergistic integration with complementary HCC therapies, maximizing patient outcomes.
Clinical Development
Led by Professor Zhi-ping Yan, Director of the Department of Interventional Radiology at Zhongshan Hospital, Fudan University, and National Chairman of the Chinese Society of Interventional Radiology.
▸ Obtained approval from the National Medical Products Administration (NMPA)
C-Wave®
Vascular Catheter System
PRODUCT CENTER
Developed independently by Zhonghui Medical, a subsidiary of H&H Medical, the C-Wave® vascular catheter system is indicated for the treatment of intravascular calcified lesions and can be used either as a standalone therapy or in combination with stents.C-Wave® employs a breakthrough "adaptive shockwave technology" that follows the principle of “gentle on soft tissue, forceful on calcified structures”. This innovative mechanism enables targeted fracturing of calcium buildup from within the vessel wall while preserving surrounding soft tissues.
▸ Obtained approval from the National Medical Products Administration (NMPA)
-
2025-12-30
"One Heart, Two Treatments": One-Stop K-Clip® Tricuspid Annuloplasty Combined with Left Atrial Appendage Closure Successfully Completed at Zhongnan Hospital of Wuhan University
Hubei's First One-Stop Procedure Combining K-Clip® Tricuspid Annuloplasty and LAAC Successfully Performed at Zhongnan Hospital of Wuhan University
more >
-
2025-12-30
Pioneering Nationwide Achievement|The Fourth Affiliated Hospital of Xinjiang Medical University Independently Achieves Breakthrough in K-Clip® Minimally Invasive Tricuspid Regurgitation Repair
Recently, under the leadership of Director Jiang Shubin from the CCU Department at the Fourth Affiliated Hospital of Xinjiang Medical University, Associate Director Shen Xiangli served as the primary operator in successfully performing a K-Clip® transcatheter tricuspid valve annuloplasty for a patient with severe tricuspid regurgitation. This procedure marks the first case independently completed by a hospital team in Xinjiang.
more >
-
2025-12-30
Event Recap | Advancing Together, Reaching New Heights in Tricuspid Therapy: K-Clip® Interventional Tricuspid Valve Therapy Online Symposium
The K-Clip® Transcatheter Tricuspid Valve Annuloplasty System offers flexibility with its deliverable and retrievable design, a wide range of indications, and effectiveness in treating regurgitation caused by various etiologies.
more >
THIS IS US, LOOKING FORWARD TO THE BEST HEALTH AND THE MOST BEAUTIFUL LIFE
News Center
H&H Medical prioritizes the development and protection of intellectual property (IP), and has achieved IP Management Standard Certification. The company has filed nearly 260 patent applications to date, with over 60% covering invention patents. More than 80 patents have been granted, and 12 PCT international patent applications are under review globally, including granted PCT patents in the United States, Canada, and Europe. The company has deep expertise in high-precision metal processing, fluid dynamics simulation, and polymer material molding technologies.
Consulting Services:question@hh-healthcare.com