K-Clip® Transcatheter Tricuspid Annuloplasty System
Product Introduction
Shanghai H&H Medical Technology Co., Ltd. (referred to as H&H Medical)'s independently developed and globally first-of-its-kind K-Clip® Transcatheter Tricuspid Valve Annuloplasty System has officially received approval for market launch from the National Medical Products Administration (NMPA)!
K-Clip® has filled the domestic gap in the field of tricuspid valve intervention, becoming the first certified transcatheter product for treating tricuspid regurgitation in China. K-Clip® has also ushered in a new era of TTVA (Transcatheter Tricuspid Valve Annuloplasty) for tricuspid regurgitation globally, leading the forefront of international academic research. In the field of structural heart disease, the tricuspid valve has been slower to achieve interventional approaches due to its complex pathological mechanisms and high risks associated with traditional treatments. In recent years, with technological advancements and the accumulation of evidence-based medical data, transcatheter techniques for the tricuspid valve have gradually become a focal point in cardiovascular medicine.
inspiration and break the game

In June 2018, a wine opener, like Newton's apple, sparked the inspiration for Transcatheter Tricuspid Valve Annuloplasty (TTVA). Huihe Medical ingeniously designed the K-Clip®, which simplistically and elegantly replicates the surgical Kay's procedure through an interventional approach. This transformed the originally open-chest surgery for folding and reducing the enlarged tricuspid valve annulus into a minimally invasive procedure requiring only venous puncture. Since 2018, after evolving from hand-drawn sketches, undergoing over 100 iterations, more than 2,500 product tests, 5,000+ design drafts, and validation using over 380 live large animals, the clinically viable K-Clip® Transcatheter Tricuspid Valve Annuloplasty System was finally born.
Research and Verification
Academician Ge Junbo Team of Zhongshan Hospital Affiliated to Fudan University
On April 21, 2021, the team led by Academician Ge Junbo from Zhongshan Hospital affiliated with Fudan University, in collaboration with Professors Zhou Daxin, Pan Wenzhi, Zhang Xiaochun, Pan Cuizhen, and Li Wei, successfully performed the first human implantation of the K-Clip® transcatheter tricuspid annuloplasty system. Subsequently, 33 cases of FIM (First-in-Man study) were completed. In October of the same year, K-Clip® passed the special approval application for innovative medical devices by the Center for Medical Device Evaluation (CMDE) of the National Medical Products Administration (NMPA), entering the "green channel" of the approval process. In May 2022, K-Clip® officially launched the Tristar I single-arm target-value registration clinical study in China. In March of the following year, K-Clip® initiated the Tristar II randomized parallel-controlled registration clinical study, completing one-year follow-up by 2024. [Participating research institutions included 16 cardiac centers, such as Zhongshan Hospital affiliated with Fudan University, the Second Affiliated Hospital of Zhejiang University School of Medicine, Fuwai Hospital of the Chinese Academy of Medical Sciences, Beijing Anzhen Hospital affiliated with Capital Medical University, the First Affiliated Hospital of Zhejiang University School of Medicine, Guangdong Provincial People's Hospital, West China Hospital of Sichuan University, and Anhui Provincial Hospital.]
Evidence-Based and Exploration
The efficacy and safety of the K-Clip® transcatheter tricuspid annuloplasty system for treating severe and above tricuspid regurgitation were demonstrated through two TriStar studies. One-year follow-up data showed that K-Clip® significantly and sustainably reduced the severity of tricuspid regurgitation, promoted right heart remodeling, markedly improved cardiac function, and enhanced patients' quality of life.
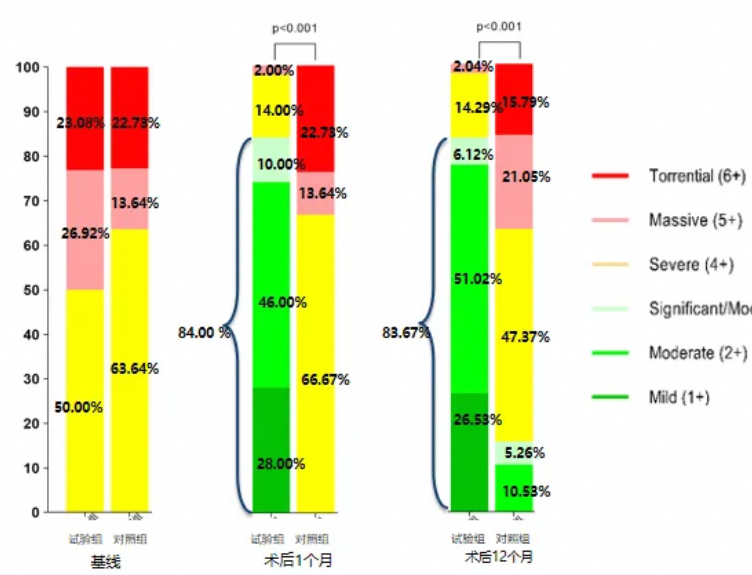
The K-Clip® transcatheter tricuspid valve annuloplasty system, as a representative TTVA procedure, is the preferred treatment for patients with severe tricuspid regurgitation primarily caused by annular dilation. Additionally, in patients with severe right heart dysfunction, K-Clip® can serve as a "bridge therapy" for TEER or TTVR procedures, preventing acute right heart failure and offering patients a chance at survival.
the past and the future
The launch of K-Clip® is not only a technological breakthrough in China but also a milestone in the global treatment of tricuspid regurgitation! The launch of K-Clip® has redefined the treatment paradigm for tricuspid regurgitation, significantly advancing the TTVA procedure. For patients, it represents a leap in survival hope and quality of life; for physicians, it liberates clinical decision-making; and for the procedure itself, it marks the beginning of an era of efficient and precise treatment. In the future, as the technology becomes more widespread and experience accumulates, it holds the potential to combine with atrial fibrillation ablation and left heart valve interventions to explore "one-stop" solutions for complex cardiac conditions.